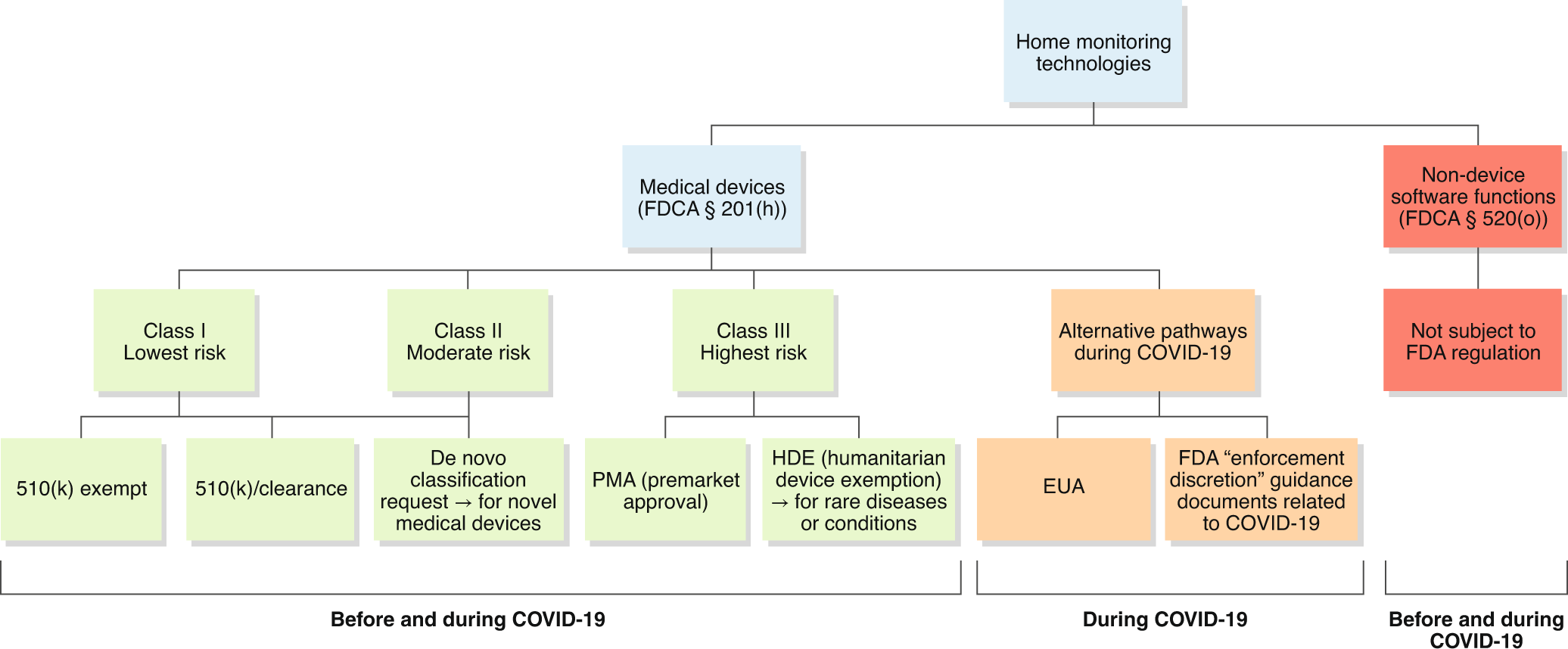
Regulatory, safety, and privacy concerns of home monitoring technologies during COVID-19 | Nature Medicine

Inhaled isoflurane via the anaesthetic conserving device versus propofol for sedation of invasively ventilated patients in intensive care units in Germany and Slovenia: an open-label, phase 3, randomised controlled, non-inferiority trial -

ISO 15223-1:2021(en), Medical devices — Symbols to be used with information to be supplied by the manufacturer — Part 1: General requirements