![B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book] B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book]](https://www.oreilly.com/library/view/essentials-of-chemical/9780134663906/graphics/f0755-01.jpg)
B. Ideal Gas Constant and Conversion Factors - Essentials of Chemical Reaction Engineering, 2nd Edition [Book]

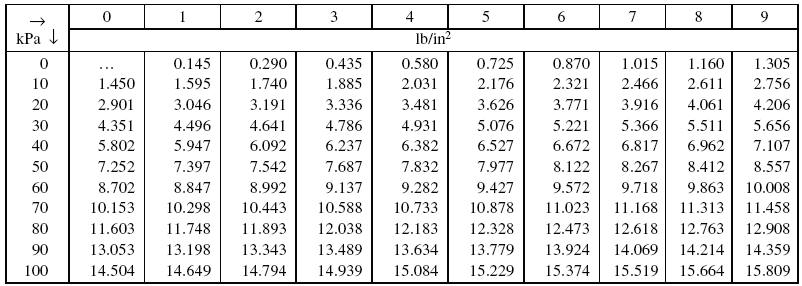
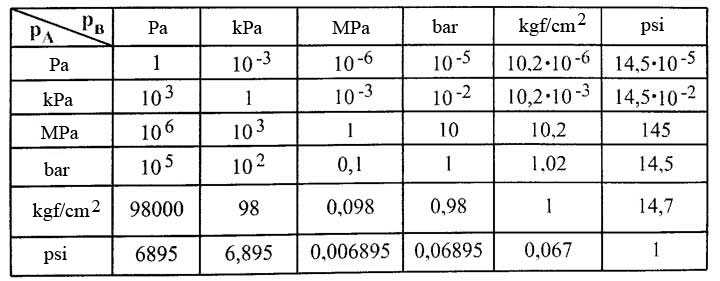
Tut 1.pdf - Tut-1 1 Convert the following readings of pressure to kPa assuming that barometer reads 760 mm of Hg(i 80 cm of Hg(ii 30 cm Hg vacuum \u03c1Hg = | Course Hero