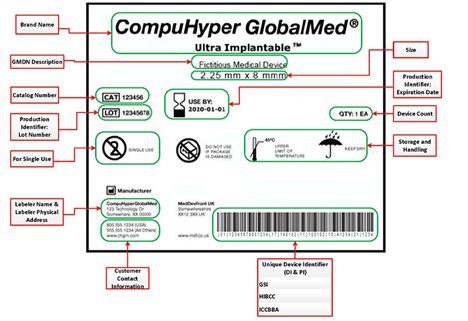
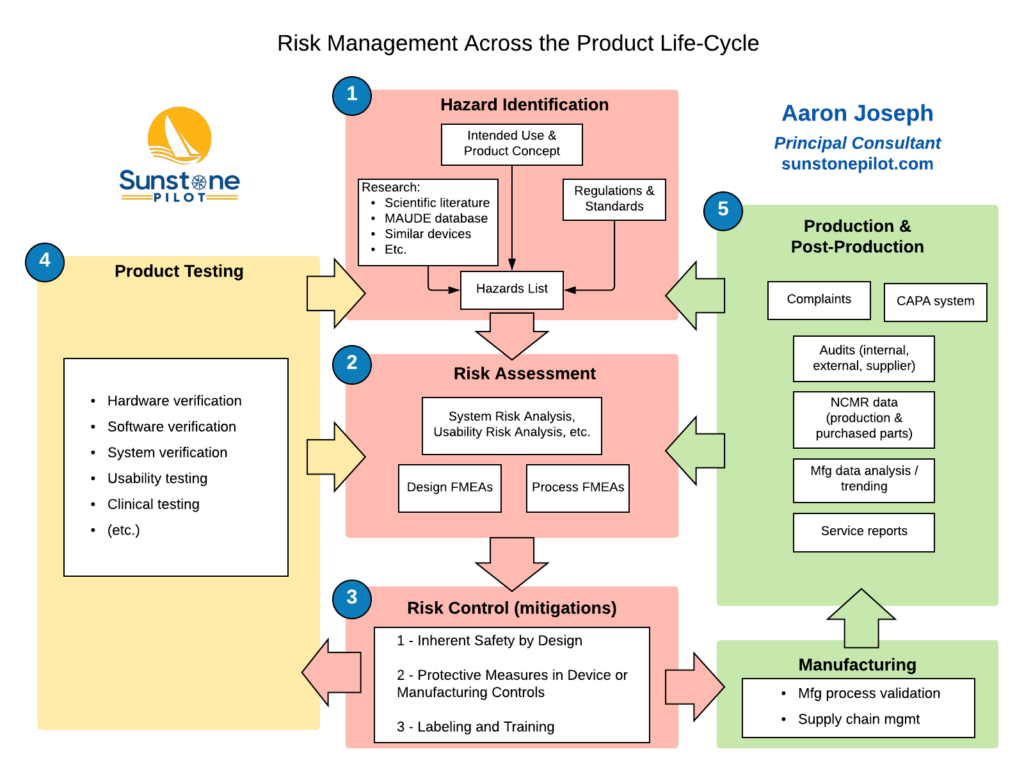
Towards a Logic-Based Extension of a Relational Software Tool for Coherent Technical Documentation of Medical Devices - In Compliance Magazine

PRISYM ID Twitterren: "PRISYM 360 provides approval and change workflow tools to ensure control over label content and compliance with the myriad regulations for #medicaldevice manufacturers and #pharma. Learn more: https://t.co/Vcpjbuqut7 https://t.co ...

Unique device identification and traceability for medical software: A major challenge for manufacturers in an ever-evolving marketplace - ScienceDirect

No more paper medical instructions - dokspot: helping MedTech companies go paperless — OneMillionSparks
![QMS for Software as a Medical Device [SaMD] Lessons Learned from a Quality Perspective - PDF Free Download QMS for Software as a Medical Device [SaMD] Lessons Learned from a Quality Perspective - PDF Free Download](https://docplayer.net/docs-images/42/17134037/images/page_2.jpg)