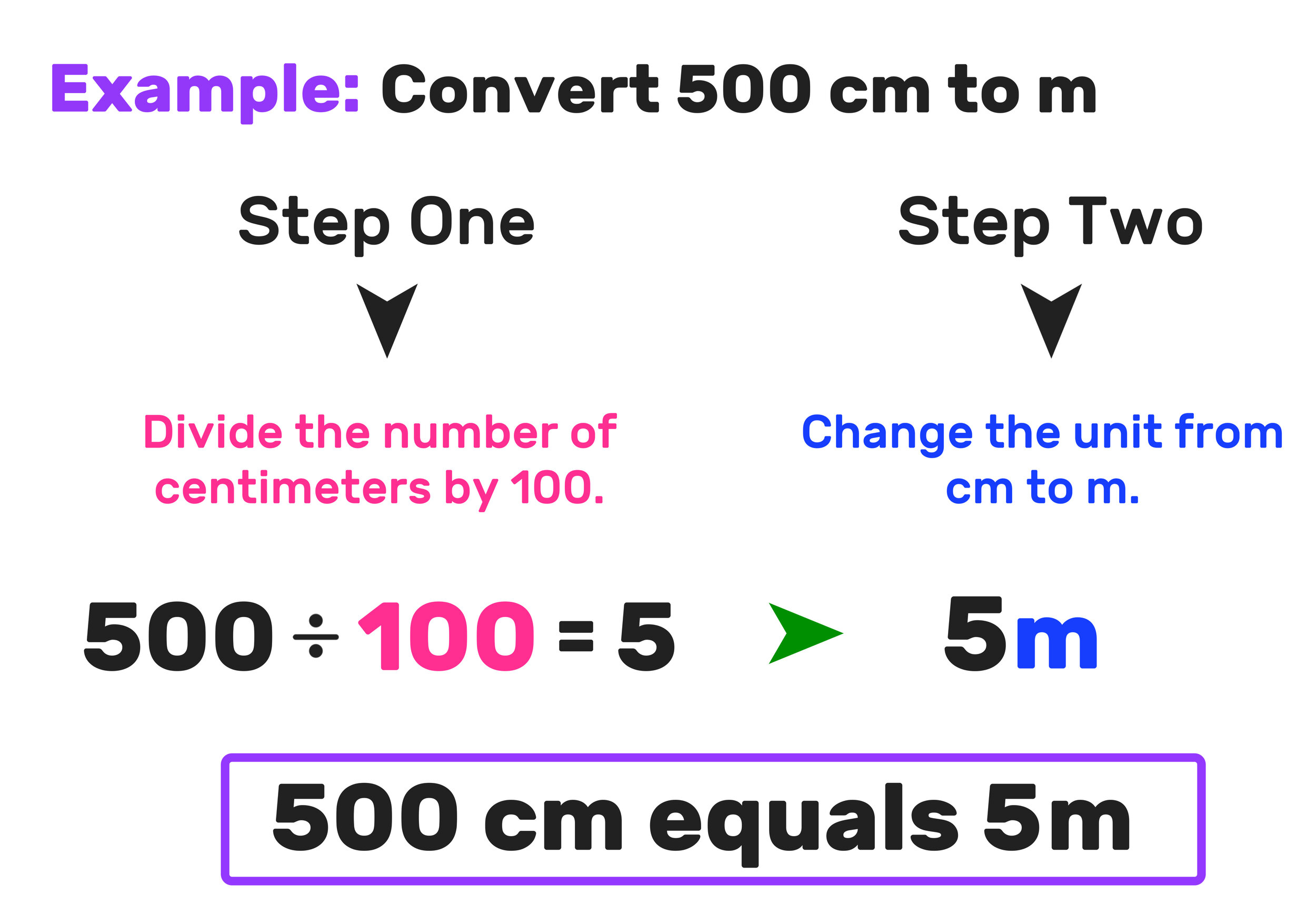
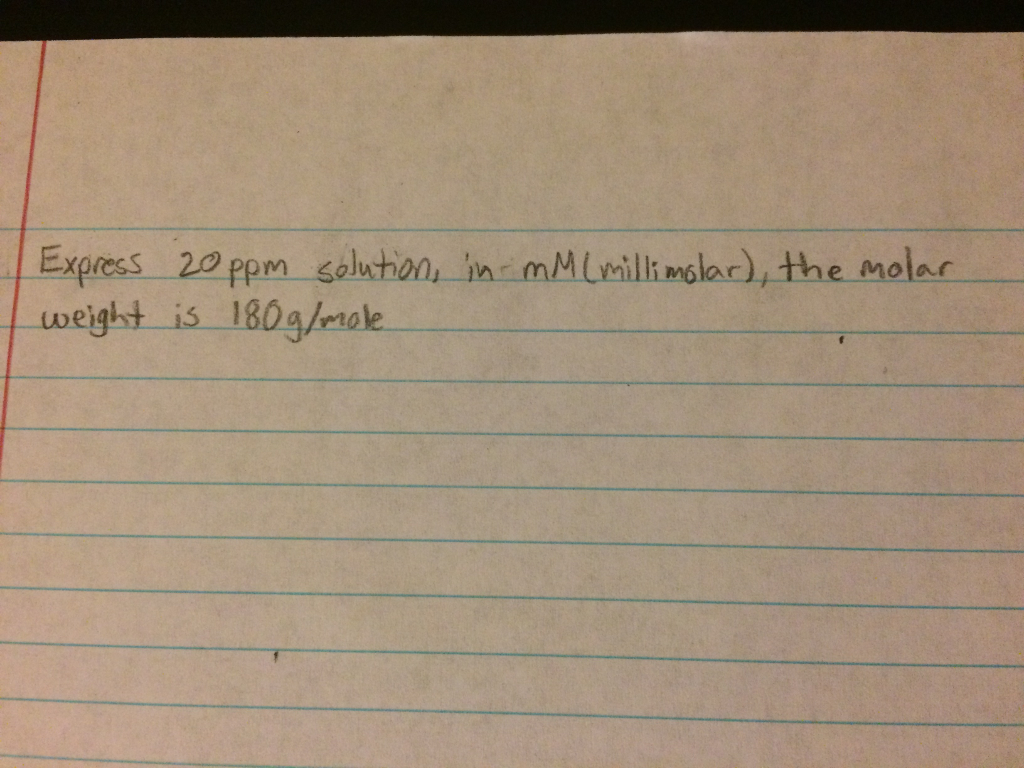Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download
![Convert millimolar [mM] to molar [M] • Molar Concentration Converter • Hydraulics — Fluids • Compact Calculator • Online Unit Converters Convert millimolar [mM] to molar [M] • Molar Concentration Converter • Hydraulics — Fluids • Compact Calculator • Online Unit Converters](https://www.translatorscafe.com/static/ucvt/img/Molar_Concentration.jpg)
Convert millimolar [mM] to molar [M] • Molar Concentration Converter • Hydraulics — Fluids • Compact Calculator • Online Unit Converters

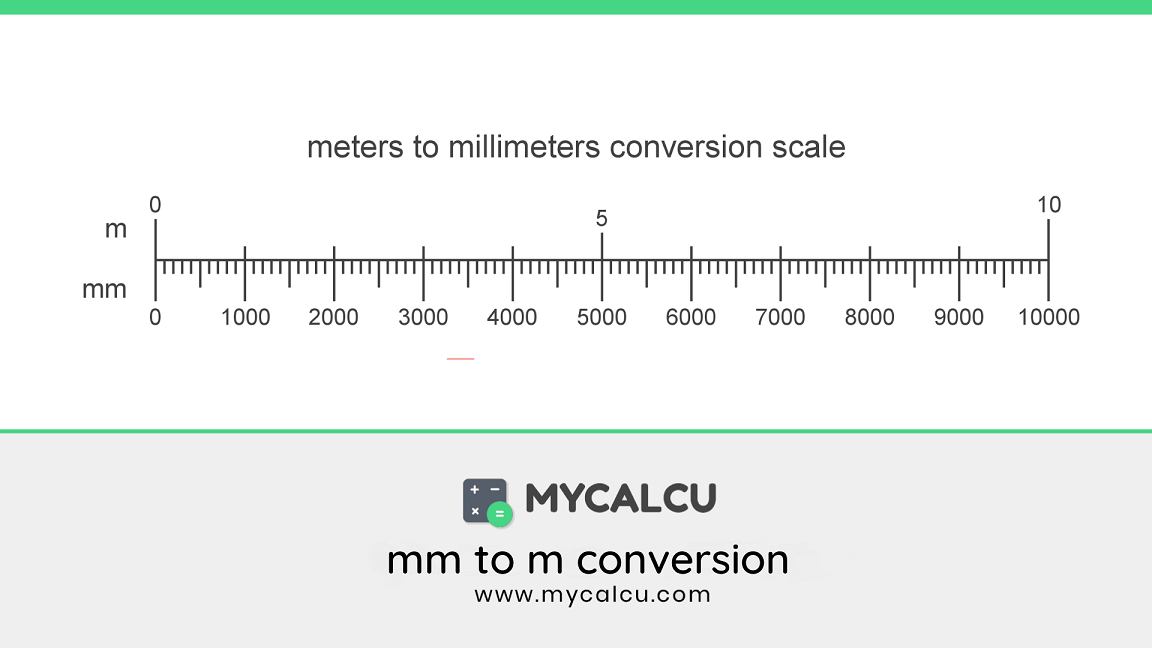




:max_bytes(150000):strip_icc()/yd2m-56a128eb5f9b58b7d0bc9742.jpg)