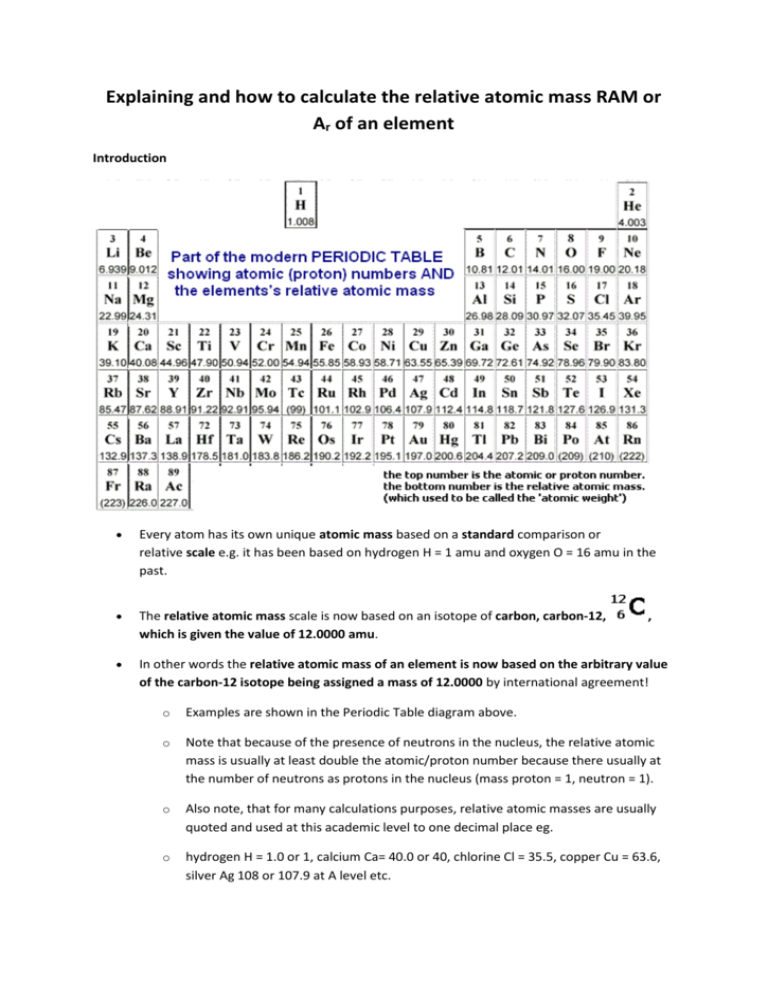
Defining how to calculate relative atomic mass of element relative isotopic mass definition gcse chemistry Calculations igcse O Level revision notes
Chlorine exists mainly as two isotopes, 37Cl and 35Cl. Which is more abundant? How do you know? - Quora